First Time Disclosure of Clinical Candidates Presentation Scheduled for April 3, 2019 at 10:35am ET
Malvern, PA, March 25, 2019 – Venatorx Pharmaceuticals announced that its President and CEO, Christopher J. Burns, Ph.D., will present during the First Time Disclosure of Clinical Candidates at the American Chemical Society (ACS) National Meeting. The presentation entitled, “Discovery of VNRX-7145: A broad-spectrum orally bioavailable beta-lactamase inhibitor (BLI) for highly resistant bacterial infections (“superbugs”)” is scheduled for Wednesday, April 3, 2019 at 10:35am ET in the Valencia Ballroom A at the Orange County Convention Center in Orlando, FL.
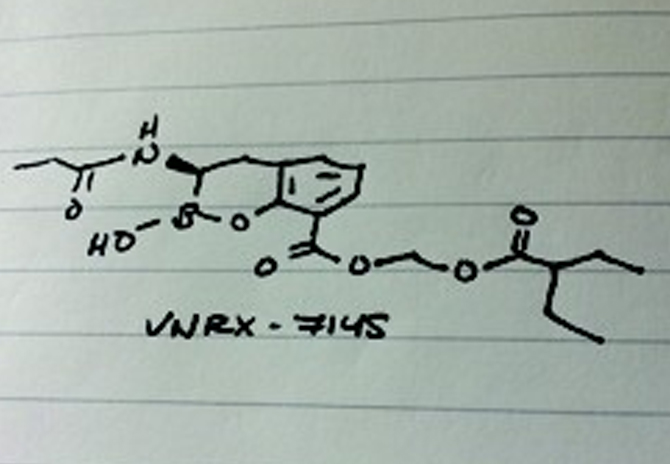
VNRX-7145 is a novel, orally-bioavailable BLI that is currently in clinical development. VNRX-7145, in combination with a known orally bioavailable cephalosporin, rescues the activity of the partner antibiotic against ESBLs and key carbapenem-resistant Enterobacteriaceae, including those expressing KPC and OXA carbapenemases. This presentation will explore the medicinal chemistry optimization to VNRX-7145 from less active, non-bioavailable lead compounds.
This project has been funded in part with Federal funds from the National Institute of Allergy and Infectious Diseases, National Institutes of Health, Department of Health and Human Services, under Contract No. HHSN272201600029C.
About Venatorx Pharmaceuticals, Inc.
Venatorx is a private pharmaceutical company that is focused on the discovery and development of novel anti-infectives to treat multi-drug-resistant bacterial infections and hard-to-treat viral infections. Founded in 2010, Venatorx has built a world-class in-house R&D organization that has filed over 100 patents spanning multiple research programs. Venatorx has received significant funding awards from the National Institute of Allergy and Infectious Diseases (NIAID) of the National Institutes of Health (NIH), Wellcome Trust, CARB-X, and the Defense Threat Reduction Agency (DTRA) as well as private equity investments from Versant Ventures, Abingworth, and Foresite Capital.
The Company’s most advanced development-stage product is VNRX-5133, an injectable beta-lactamase inhibitor (BLI) that features selective and potent in vitro activity against both serine- and metallo-beta-lactamases, including OXA, KPC, NDM, and VIM. Venatorx believes that VNRX-5133, in a fixed combination with the fourth generation cephalosporin, cefepime, has the potential to provide a valuable broad-spectrum treatment option to meet unmet medical need in patients with infections due to carbapenem-resistant pathogens including carbapenem-resistant Enterobacteriaceae (CRE) and carbapenem-resistant Pseudomonas aeruginosa (CRPA), suspected polymicrobial infections caused by both gram-negative and gram-positive susceptible pathogens, and engineerable MDR bioterror pathogens such as Burkholderia spp. and Salmonella spp. Early clinical studies of cefepime/VNRX-5133 have been completed and Venatorx intends to initiate Phase 3 pivotal trials during the second quarter 2019.
Venatorx’s second development-stage product in clinical development is VNRX-7145, which is an orally bioavailable BLI that in combination with a known orally-bioavailable cephalosporin, rescues the activity of the partner antibiotic against ESBLs and key carbapenem-resistant Enterobacteriaceae, including those expressing KPC and OXA carbapenemases. Additionally, Venatorx has a broad pipeline of preclinical programs including a novel class of Penicillin-Binding Protein (PBP) inhibitors that are impervious to beta-lactamase-driven resistance, and novel antiviral agents targeting Hepatitis B Virus. For more information, please visit www.venatorx.com.
